How much does a 135 lbs patient weigh in kilograms?
Answer: 61.235 kg Explanation: To convert pounds to kilograms, you can use the conversion factor where 1 pound is approximately 0.453592 kilograms. Thus, to convert 135 pounds to kilograms: \[ 135 \, \text{lbs} \times 0.453592 \, \text{kg/lb} \approx 61.235 \, \text{kg} \]
How much is 71 inches in feet?
Answer: \( 5.9167 \) feet (approximately \( 5 \) feet \( 11 \) inches) Explanation: To convert inches to feet, divide the number of inches by \( 12 \) (since there are \( 12 \) inches in a foot). Steps: Divide \( 71 \) by \( 12 \): \[ \frac{71}{12} \approx 5.9167 \] The whole […]
How many ounces are in 1/2 cup?
Answer: 4 ounces Explanation: There are 8 ounces in 1 cup, so to find the number of ounces in 1/2 cup, you divide 8 by 2, which equals 4 ounces.
How many milliliters are in a milligram?
Answer: 1 milligram (mg) is equal to 0.001 milliliters (mL) for water at standard temperature and pressure. Explanation: The conversion between milligrams and milliliters depends on the density of the substance. For water, the density is 1 g/mL, meaning 1 g (1000 mg) occupies 1 mL. Therefore, to convert milligrams to milliliters for water, you […]
The formula to calculate the area of a circle from its circumference is: A = c² / 4π
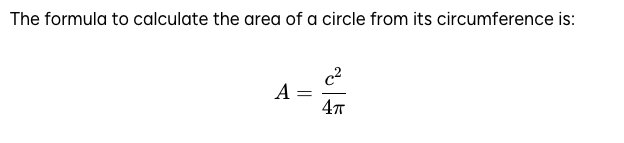
Answer: \( A = \frac{c^2}{4\pi} \) Explanation: The formula provided relates the area \( A \) of a circle to its circumference \( c \). The circumference of a circle is given by \( c = 2\pi r \), where \( r \) is the radius. The area of a circle can also be expressed […]
How many cups are equivalent to 2 pounds?
Answer: 4 cups Explanation: The conversion from pounds to cups depends on the substance being measured. For granulated sugar, 1 pound is approximately equal to 2 cups. Therefore, for 2 pounds, it would be \( 2 \times 2 = 4 \) cups.
Why is the value of k taken as 9×10, 10 is square of 9?
Answer: The value of \( k \) is taken as \( 9 \times 10 \) because it represents a specific relationship or calculation in a given context, possibly related to a problem involving area, volume, or another mathematical concept. Explanation: In many mathematical problems, particularly those involving geometric figures or physics, constants like \( k […]
When do we use R constant as 0.0821 and 8.
Answer: The gas constant \( R \) can be used in different forms depending on the units of pressure, volume, and temperature. Explanation: The value of \( R \) is used in the ideal gas law \( PV = nRT \), where: \( R = 0.0821 \, \text{L} \cdot \text{atm} / (\text{mol} \cdot \text{K}) \) […]
List several activities during which the near point is important.
Q1: Activities involving reading or writing Q2: Activities involving using a computer or smartphone Q3: Activities involving crafting or detailed handiwork Q4: Activities involving playing musical instruments Answer: The near point is important in various activities that require close visual focus. Explanation: The near point refers to the closest distance at which the eye can […]
According to the rules of osmosis, at what point will a system reach a state of equilibrium? A system will reach equilibrium when the __________________ on both sides of a water-permeable membrane are equal. Answers: a) concentration of solute molecules b) number of solute molecules c) number of solvent molecules d) volume of water
Answer: a) concentration of solute molecules Explanation: A system will reach equilibrium when the concentration of solute molecules is equal on both sides of a water-permeable membrane. At this point, there is no net movement of water or solute molecules across the membrane, resulting in a stable state.
